By Vanessa and Miles
Chlorofluorocarbon otherwise known as CFCs were originally used and created in 1928 because they were a safe alternative to dangerous chemicals such as ammonia which used to be used in refrigerators a refrigerant. CFC’s were considered safe because they were non- flammable and they weren’t toxic. CFCs were used in refrigerators as refrigerant and in aerosol cans so things like hairspray bottles and spray paint cans.
|
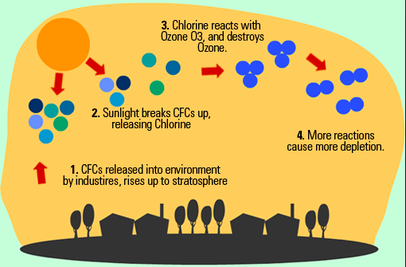
CFCs are made up of one carbon atom (C), one fluorine atom (F), and two chlorine atoms (Cl). Once CFCs get into the Ozone, Ozone depletion occurs in the stratosphere. Ozone depletions occur as ultraviolet light (UV) from the sun cause the chlorine atom to break apart and separate from the CFC molecule. Once the chlorine molecule has broken apart the atom then hits an ozone molecule (O3) which causes the chlorine (Cl) to pull away one oxygen atom (O) from the ozone molecule (O3) creating chlorine monoxide (ClO). A free oxygen (O) atom that is found in the stratosphere then will collide with the chlorine monoxide molecule (ClO) which creates another free floating chlorine molecule (Cl). This process will continue to repeat itself, always creating a free floating chlorine atom which ends up depleting the ozone. The chemical reaction for the decomposition of the oxone by the chlorine radical is Cl+O3 (g) → ClO (g) +O2 and the chemical reaction for the natural ozone decomposition by the sun is O3 + UV Radiation → O2 + O and finally the chemical reaction for the regenerated chlorine radical is O + ClO → Cl + O2.
|
The Negative impact CFCs have on the environment is that they deplete the ozone layer in the stratosphere. This is bad for the environment because the ozone layer protects us from harmful UV rays from the sun. The more ozone that disappears means the more UV rays that reach the earth which causes global warming which cause glaciers to melt, ice is melting all around the world which is affecting animals like polar bears who rely on the ice to drowned, and the melting of all the ice has caused the sea levels to rise. People used CFCs for almost 50 years without realizing how harmful they were to the ozone layer. It wasn’t until the early 1970’s that people decided to look at the effects that CFC’s were having in the ozone. They started to notice that in 1985 that above antarctica the ozone layer was becoming thinner. This caused people to panic because people thought that the decrease in ozone would increase the chances of skin cancer because more UV rays were getting through.
|
|
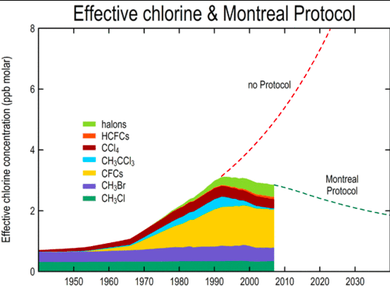
The Canadian Government Regulation put in place was the Montreal Protocol. The Montreal Protocol was an international treaty that was created in order to protect the ozone layer by putting a stop to the use of CFCs and on September 16th, 1987 it was signed. By January 1st 1987 it was put into place and since then the ozone has began to recover and fix itself.
|
Alternative technologies used nowadays in place of CFCs are Hydrofluorocarbons (HFCs). HFC’s and CFC’s are basically the same thing except for one crucial difference. HFCs don’t use chlorine which means that if they get into the ozone it isn't that big of a deal because the chemical reaction that was occurring between the chlorine and ozone does not occur. That means it is safe to use HFC’s as a substitute for CFCs so they are now used in refrigerators and air conditioners as coolants and they are also used in aerosol cans.