Asbestos, Carbon tetrachloride, and trichloroethylene are all examples of chemicals that are included on the Toxic Substance List of Canada.
|
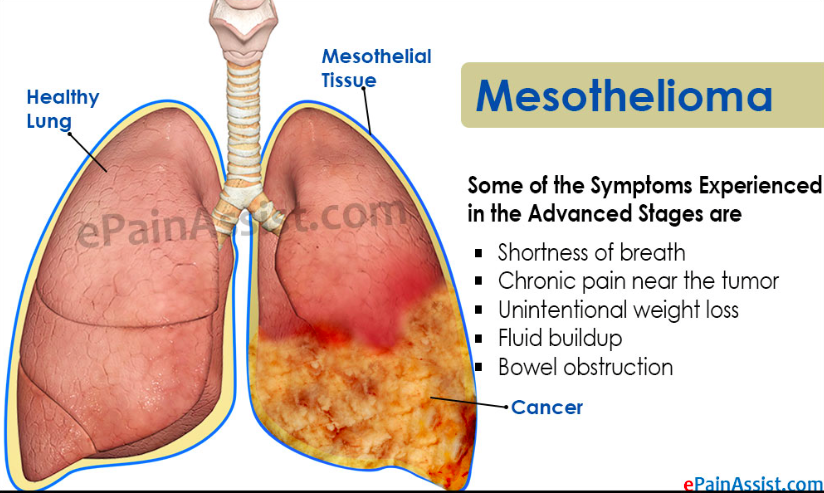
People started realizing asbestos effects in the 1930’s and By the 1960s, serious diseases like mesothelioma were firmly established as being caused by asbestos exposure. Asbestos is now banned or heavily regulated because of its carcinogenic and toxic properties. The World Health Organization and the International Labor Organization have condemned its use. Commercial trichloroethylene contains stabilizers, which may contribute to toxicity. Can cause dry, red, cracked skin (dermatitis) following skin contact. Conclusions cannot be drawn from the limited studies available. May harm the nervous system. Symptoms may include headaches, fatigue, memory loss, irritability, depression and reduced ability to think or reason. The nerves of the face and head (cranial nerves) may be affected by long-term exposure to trichloroethylene or chemicals formed when it decomposes. After exposure to high levels of carbon tetrachloride, the nervous system, including the brain, is affected. Such exposure can be fatal. The immediate effects are usually signs of intoxication, including headache, dizziness, and sleepiness perhaps accompanied by nausea and vomiting. These effects usually disappear within 1-2 days after exposure stops. In severe cases, stupor or even coma can result, and permanent damage to nerve cells can occur.
Alternatives to asbestos include polyurethane foams for any structural use as well as insulation. Flour fillers can be used for filling structural cracks and crevices in buildings. Cellulose fiber is one of the most popular alternatives to asbestos, made from finely shred newsprint. Chemically treated to increase fire resistance and reduce mold, cellulose fiber is generally made of 85 percent recycled content, making it a viable green option. It is good for use in commercial or residential buildings.
|
The human health effects from long-term unsafe asbestos exposure are well documented. Asbestos fibres are easily inhaled and carried into the lower regions of the lung where they can cause fibrotic lung disease and changes in the lining of the chest cavity. Acute inhalation and oral exposures to high levels of carbon tetrachloride have been observed primarily to damage the liver (swollen, tender liver, changes in enzyme levels, and jaundice) and kidneys (nephritis, nephrosis, proteinuria) of humans. Depression of the central nervous system has also been reported. Symptoms of acute exposure in humans include headache, weakness, lethargy, nausea, and vomiting. Delayed pulmonary edema (fluid in lungs) has been observed in humans exposed to high levels of carbon tetrachloride by inhalation and ingestion, but this is believed to be due to injury to the kidney rather than direct action of carbon tetrachloride on the lung. Acute animal exposure tests in rats, mice, rabbits, and guinea pigs have demonstrated carbon tetrachloride to have low toxicity from inhalation exposure, low to moderate toxicity from ingestion, and moderate toxicity from dermal exposure. No information is available on the acute effects of trichloroethylene in humans from inhalation or oral exposures. Studies on dermal exposure to trichloroethylene in humans have reported stinging and burning sensations and transient whitening of the skin. Animal studies have reported effects on the liver, kidney, and CNS from acute inhalation and oral exposure to trichloroethane. Tests involving acute exposure of mice and rats have shown trichloroethane to have moderate and high acute toxicity from inhalation and oral exposure.
It was deemed that the scientific basis of their negative effect on the environment and/or human life or health had been sufficiently demonstrated and that determination of their negative effect was consistent with the criteria set out in CEPA 1988. Therefore, the Government of Canada, on the recommendation of the Ministers of Environment and Health, decided that no further assessment would be required CEPA 1988. Substances for which an assessment was conducted in accordance with the criteria set out in CEPA 1988 to determine whether or not they were toxic or capable of becoming toxic. In respect of these substances, a determination of "toxic" under CEPA 1988 was made, and the Ministers of Environment and Health recommended their addition to the List of Toxic Substances in Schedule 1 of CEPA 1988.
|